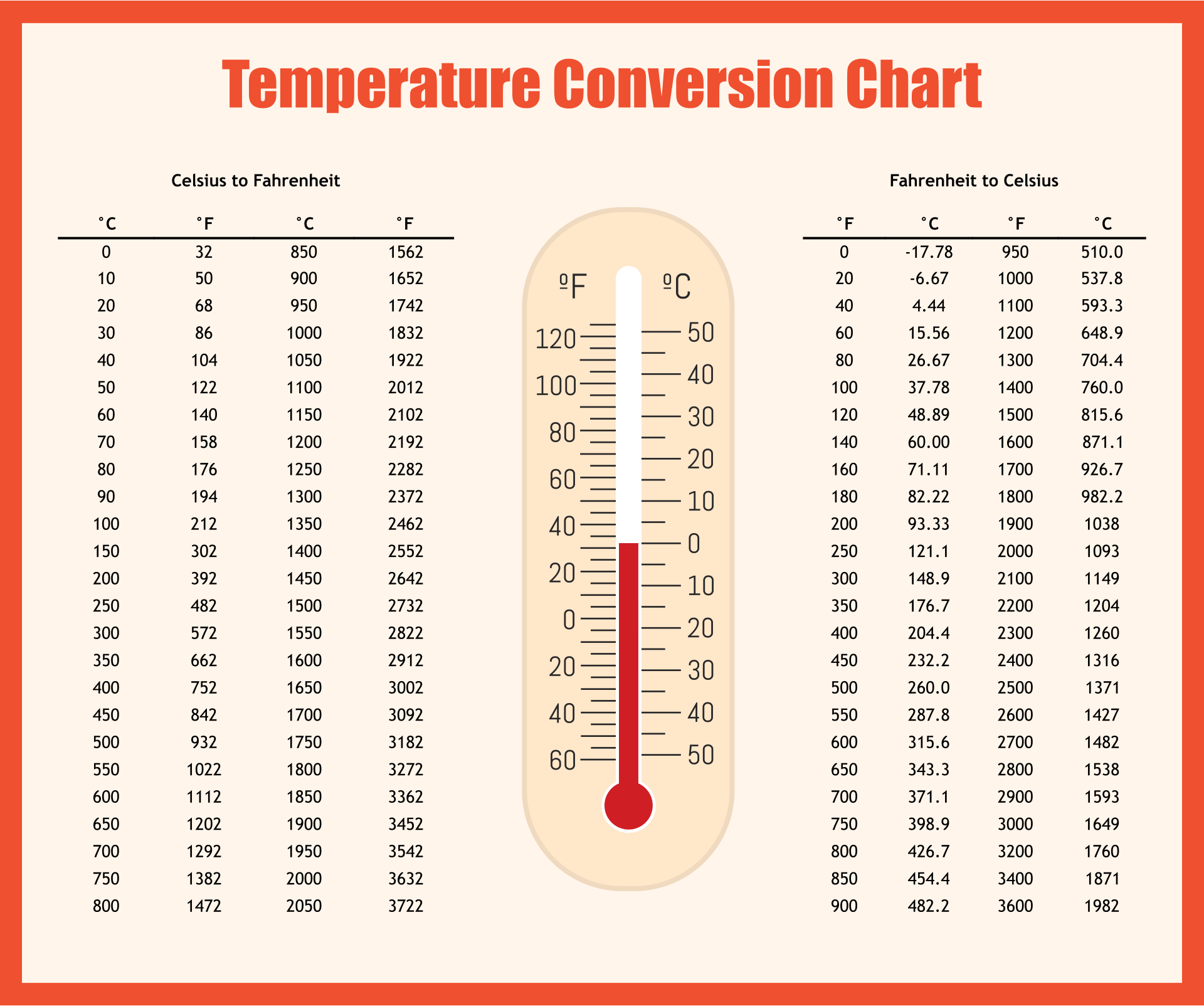
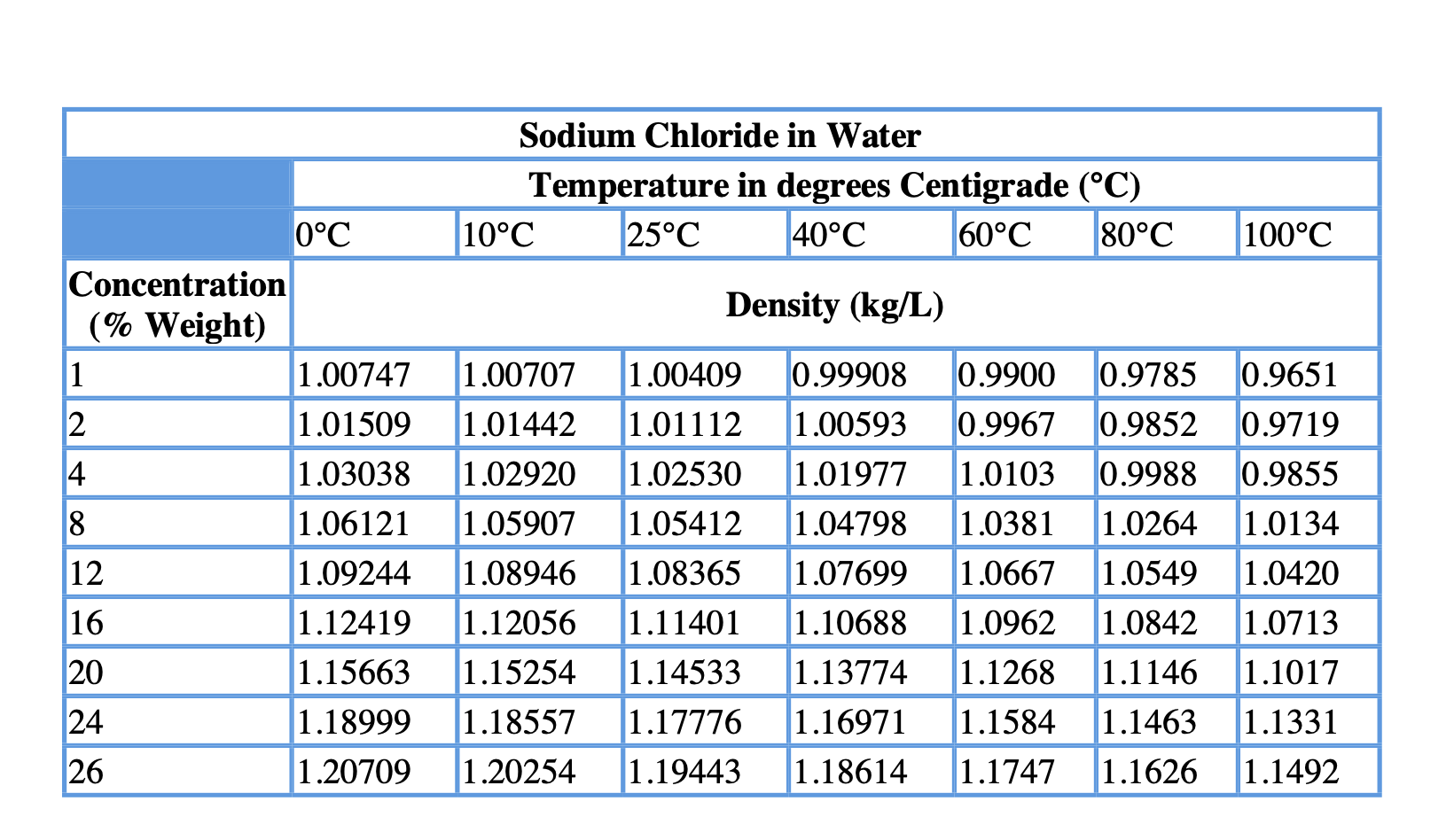
The total number of moles present in the solution will be Use glucose and water's respective molar masses to determine how many moles of each you have This means that all you have to do is determine the mole fraction of water in the solution.Īs you know, mole fraction is defined as the number of moles of a component of a solution divided by the total number of moles present in that solution. In your case, you know that the vapor pressure of pure water at is equal to #23.8# torr.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
